Excitons do the Long Jump – published in Physical Review Letters
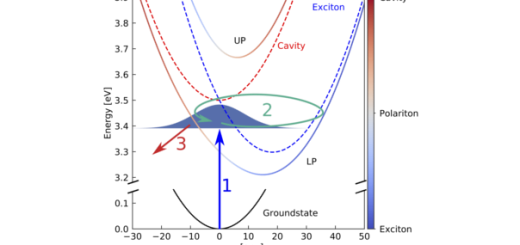
 When light is absorbed in organic materials, its energy is converted into excitons (electronic excitations) that normally move around by “hopping” from molecule to molecule. This is important in photosynthesis or solar cells, for example. There, the energy of the absorbed sunlight has to be brought to a “reaction center”, where it can be used to power chemical or electrical processes. In many organic materials, energy transport is inefficient because of the large amount of disorder and motion of the molecules. The theoretical work, carried out by IFIMAC researchers Johannes Feist and Francisco J. Garcia-Vidal, shows that an extraordinary increase of the transportation efficiency (or “exciton conductance”) can be achieved by placing two mirrors on both sides of the organic material. The excitons can then not only hop from molecule to molecule, but also transfer their energy back into light trapped between the mirrors. When this happens quickly enough, the exciton and light behave as an effective new particle with hybrid properties borrowed from both constituents. In this regime of “strong coupling”, energy transferred into the system at one spot can efficiently “jump” over a large distance, bypassing the disordered collection of molecules.
When light is absorbed in organic materials, its energy is converted into excitons (electronic excitations) that normally move around by “hopping” from molecule to molecule. This is important in photosynthesis or solar cells, for example. There, the energy of the absorbed sunlight has to be brought to a “reaction center”, where it can be used to power chemical or electrical processes. In many organic materials, energy transport is inefficient because of the large amount of disorder and motion of the molecules. The theoretical work, carried out by IFIMAC researchers Johannes Feist and Francisco J. Garcia-Vidal, shows that an extraordinary increase of the transportation efficiency (or “exciton conductance”) can be achieved by placing two mirrors on both sides of the organic material. The excitons can then not only hop from molecule to molecule, but also transfer their energy back into light trapped between the mirrors. When this happens quickly enough, the exciton and light behave as an effective new particle with hybrid properties borrowed from both constituents. In this regime of “strong coupling”, energy transferred into the system at one spot can efficiently “jump” over a large distance, bypassing the disordered collection of molecules.
Reference:
- Extraordinary Exciton Conductance Induced by Strong Coupling, Johannes Feist and Francisco J. Garcia-Vidal, Phys. Rev. Lett. 114, 196402, (2015). [URL]